
News




INFORMATION TECHNOLOGY : A Risk-Based Approach for Computer Systems Validation SITES : Trial Monitoring - Source Document Verification REGULATORY : FDA's Guidance on Patient-Reported Outcomes ALSO IN THIS ISSUE : FDA's Oversight Capabilities Increased, Influenza Warning Issued, Cloud Computing: A Reality?, Guidance Says Focus on the Patient

eClinical Trials Collaboration Challenge Integrated Technology Manage Trial Master Files via Investigative Portals Directory
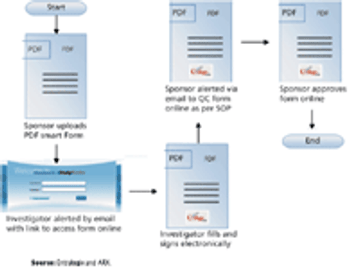
Automated eTMF solutions reduce costs, improve productivity, and enhance data management.
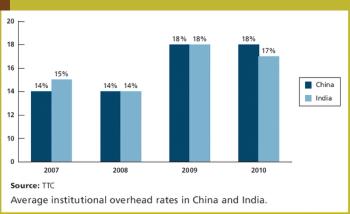
Asia, Latin American, and Eastern Europe are increasingly attractive geographies in which to conduct clinical research.

Though they are the largest minority in the country, Hispanics remain underrepresented.
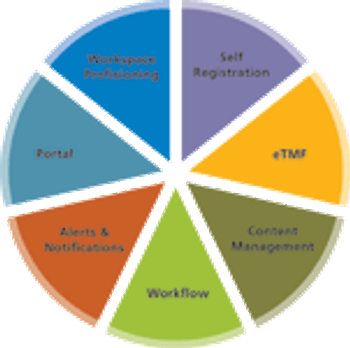
The seven elements of an integrated clinical collaboration platform.


Merck's cardiovascular drug vorapaxar study was halted.

Considerations in implementing a risk-based framework for computer systems validation.


Industry news focusing on the people and organizations who work in the clinical trials profession.














