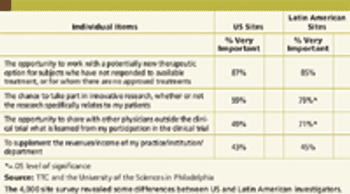
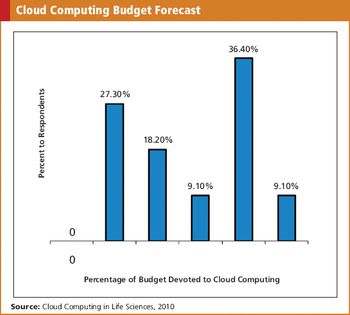
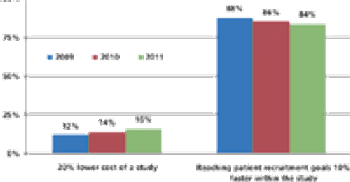
CRO/SPONSOR : Clinical Contracting Efficiency Can Save Millions REGULATORY : Common Technical Document Development - A Partnership Approach Also in this issue : FDA Looks to Collaborate Overseas, EU Reviews, Clinical Trial Rules, Meaningful Web, Greater Trial Insight